Cytomegalovirus in Pregnancy
Authors
INTRODUCTION
Cytomegalovirus (CMV), a member of the herpes virus group, is the most common cause of congenital viral infection1,2 and the most common infectious cause of developmental delay and sensorineural hearing loss in the United States.3 Worldwide, 1% of all live-born infants are infected with CMV. CMV is ubiquitous, highly species specific, and, like other members of its family, infects almost all human beings at some point during their lives.4 The age at acquisition varies according to geographic and socioeconomic factors resulting in large differences in prevalence among groups.
The natural history of CMV infection is complex and characterized by lifelong latency punctuated by episodes of recurrent infection following a primary infection. After a primary infection, viral excretion from several different sites may persist for weeks to years before the virus becomes latent. Episodes of recurrent infection with renewed shedding often represent reactivation of latent virus but also can be caused by reinfection by an antigenically different strain of CMV. Regardless of stage of infection, most episodes of CMV infection are asymptomatic and do not pose significant health threat to immunocompentent hosts. However, maternal infection during pregnancy can cause serious, permanent sequelae in the fetus. Despite the potentially devastating nature of congenital infection, there is no effective treatment.
MICROBIOLOGY
Cytomegalovirus is the largest member of the herpes virus family, which also includes herpes simplex types 1 and 2, Epstein-Barr virus (EBV), and varicella-zoster. Its genome is composed of 240 kilobases of double-stranded DNA that code for greater than 100 proteins.5 The virus consists of a 64-nm core enclosed by a 110-nm icosahedral capsid composed of 162 capsomeres. A loosely applied lipid bilayer envelope surrounds a poorly defined tegument that, in turn, surrounds the viral capsid. The mature, enveloped viral particle is approximately 200 nm in diameter.6 Many antigenically diverse strains of the virus exist, but cross-reactivity is common. Genomic studies using digestion with restriction endonucleases have shown an average homology of 80% between strains; however, thousands of different strains are recognized.7 Latent CMV has been found in monocytes, lungs, spleen, kidneys, secretory glands,8 and the cervix.9
The triphasic replication cycle of CMV, restricted to human cells, is characterized by immediate-early, early, and late phases.10 Akin to other herpes viruses, cascade synthesis of mRNA and proteins correlated with the three phases of the cycle (named α, β, γ, respectively) closely control the temporal expression of the viral genome.7,11 The replication cycle is relatively long, requiring approximately 96 hours for release of progeny virus from cells infected in vitro. Replication of viral DNA takes place within the nucleus of the infected cell, and the envelope is acquired by budding through the nuclear membrane into a cytoplasmic vacuole.12
CMV cytopathology is characterized by cell enlargement and rounding, a basophilic intranuclear inclusion, and an eosinophilic paranuclear inclusion, which often gives infected cells a characteristic “owl's eye” appearance.12 In addition to productive infection that leads to the release of active viral particles, CMV can establish a latent infection in which viral DNA persists in the host without active replication.
Growth of the virus in tissue culture can be difficult and time-consuming. Human fibroblasts are the only cells in which CMV reliably grows in vitro, despite the virus's tropism for epithelial cells in vivo.2 The characteristic cytopathic effect develops within hours to weeks after inoculation depending on the amount of CMV present in the specimen.13
IMMUNOLOGY
Host control of CMV replication involves both nonspecific and specific defense mechanisms. Induction of natural killer cells and induction of interferon immediately after CMV infection appear to be the most important aspects of the nonspecific host defense against the virus. The most important aspects of the specific immune defense against CMV are cell mediated, as functioning T lymphocytes are necessary to control the latent virus.14 Humoral immune responses to CMV do not appear to be important defenses against infection. For example, the fetus can be infected by intrauterine transmission of CMV in a woman known to have antibodies present before conception and the neonate can be infected by CMV in breast milk despite the presence of passively acquired maternal antibodies. Therefore, the presence of antibody should not be considered a measure of immunity but rather a marker of previous infection with CMV. Although specific antibodies cannot prevent CMV infection, they do seem to moderate its pathogenicity.7
TRANSMISSION
The persistence of a large reservoir of CMV in human populations in the absence of any known nonhuman vectors represents a remarkably successful form of parasitism. This success is because of the lengthy persistence of viral excretion after primary infection, the multiple sites of viral excretion, the intermittent reactivation and excretion of virus, and the relatively indolent course of infection, which allows the immunocompentent host to live indefinitely with the virus. Direct or indirect horizontal transmission occurs primarily by contact with saliva, urine, cervical or vaginal secretions, semen, breast milk, or blood.15,16,17
Close or intimate person-to-person contact usually is necessary for viral transmission. Accordingly, sexual transmission, breastfeeding, and use of day care have been implicated in the spread of CMV. Seropositivity is higher among persons with multiple sexual partners and histories of sexually transmitted diseases.18 Breastfeeding is a potential source of transmission to the newborn because seropositive women can secrete the virus in breast milk.19 Day care centers contribute to the spread of CMV by promoting close contact of children, many of whom are excreting the virus.20,21,22 Toddlers, in particular, are a key source of infection. Adler and coworkers23 found 20% to 40% of toddlers attending a day care facility to be excreting CMV. Horizontal transmission of CMV from child to child occurs by transfer of virus in saliva on hands and toys.24,25
Perinatal transmission of CMV occurs through several mechanisms. Transplacental transfer of virus in utero and fetal exposure to CMV secreted in the vagina during passage through the birth canal at delivery are the major modes of vertical transmission. Postnatally, infection can be acquired through breast milk or through direct person-to-person contact.26 In contrast to in utero (congenital) infection, peripartum infection (either intrapartum or postpartum) does not pose serious harm to the development of the neonate, except in babies weighing less than 1500 g.27
EPIDEMIOLOGY
The prevalence of antibody to CMV increases with age, but the patterns of acquisition vary widely among populations according to geographic and socioeconomic factors. In general, the prevalence of CMV infection is higher in developing countries and among those with a low socioeconomic status. In the United States, 40% to 60% of children of low socioeconomic status are infected by age 5 years, and by age 20 to 25 years, almost 80% to 100% have acquired the infection.28 In comparison, only 40% to 60% of middle-upper class adults are CMV seropositive.29 Infection is continually acquired throughout life such that by age 60 to 70 years, the majority of individuals are seropositive for CMV regardless of socioeconomic status.28
The percentage of serologically susceptible women of reproductive age also varies by geography and socioeconomic status, with lower susceptibility rates in Sweden and higher rates in the United Kingdom and the United States. On average, susceptibility rates range from 28% to 48% among middle-income populations of developed countries.30,31,32,33,34 Maternal seropositivity has also been correlated with multigravidity, older age, first pregnancy before age 15, and a greater total number of sex partners.18 Prospective studies of women in the United States indicate that the rate of CMV acquisition for women of childbearing age of middle-to-upper socioeconomic status is approximately 2% per year compared to 6% among women of low socioeconomic background.35 Because the highest rate of seroconversion occurs between the ages of 15 and 35 years and a significant portion of the reproductive population is susceptible, the possibility that primary infection coincides with pregnancy is relatively high.36
Studies of CMV acquisition during pregnancy in locales as diverse as Texas, Alabama, Britain, and Sweden yielded similar rates of 0.7% to 3.7% for prenatal primary infection among susceptible women.29 Risk factors for seroconversion during pregnancy include young maternal age, having young children at home,37 having children ages 12 to 18 years at home,29 and nonwhite race.38
Of particular concern is the risk to seronegative mothers who have children in day care as well as seronegative day care workers who become pregnant, because high rates of CMV transmission have been documented in day care settings.39 Studies have found that parents of children in day care are more likely to seroconvert than parents whose children are not in day care.23,40 Adler and coworkers41 found that 30% of mothers of toddlers infected in day care became infected themselves within 1 year compared with only 3% of mothers of uninfected toddlers. Therefore, having children younger than 3 years of age with CMV infections acquired at day care is an important risk factor for maternal acquisition of CMV. Day care workers also are at increased risk of CMV acquisition. Adler and coworkers42 reported annual seroconversion rates of 11% for day care workers compared with 2.2% for hospital employees matched for age, race, and marital status. For women working with children younger than 3 years of age, the risk of primary infection increases to 30% per year.39
Hospital workers in contact with neonates, children, or immunocompromised patients also may be at increased risk for primary CMV infection. Although several studies have found trends toward higher seroconversion rates among hospital workers in contact with high-risk patients, a clear relationship is not established. Yeager43 documented seroconversion rates of 4.1% and 7.7% per year for neonatal nurses and pediatric nurses, respectively, compared with 0% for nonhospital employees. Similarly, Friedman and coworkers44 found seroconversion rates among staff at a pediatric hospital to be higher among employees with patient contact compared with those without patient contact. However, two other cohort studies failed to find significantly different seroconversion rates among neonatal nurses, other healthcare workers, and pregnant women in the community.45,46 Given the sum of the data, pediatric or transplant nursing should not necessarily be considered an occupational risk for CMV infection.
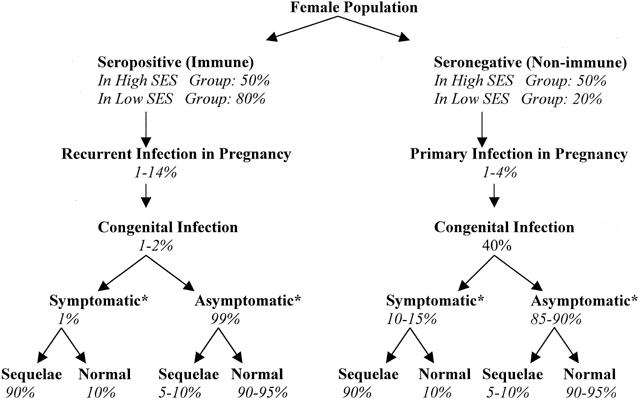
The incidence and severity of fetal infection varies according to whether the mother experiences primary or recurrent CMV infection during pregnancy. Figure 1 provides a flow diagram that highlights the higher attack rate, the higher rate of symptomatic congenital infection, and the worse prognosis of congenital infection among infants exposed to primary compared with recurrent maternal infection. Approximately 1% to 4% of seronegative pregnant women experience primary CMV infection during pregnancy.29 The rate of fetal infection after primary maternal infection is 30% to 50%.33,35 At birth, 10% of these congenitally infected infants show signs of CMV inclusion disease (CID). The remaining 90% of these congenitally infected infants are asymptomatic at birth; however, even 10% to 15% of those asymptomatic at birth experience long-term sequelae.3,47
|
By contrast, recurrent maternal infection during pregnancy, occurring at a rate of 1% to 14% among seropositive women, is more frequent than maternal primary infection during pregnancy. Unlike primary maternal infection, however, only 0.2% to 2% of recurrent infections cause congenital infection, and the neonatal sequelae of these congenital infections tend to be less severe. More than 99% of infants congenitally infected after recurrent maternal disease are asymptomatic at birth. Asymptomatic neonates may still experience long-term sequelae but at a lower incidence (5% to 10%) than those congenitally infected during primary maternal infection.3
MATERNAL INFECTION
Symptoms and Signs
For most immunocompentent adults, primary or recurrent infection with CMV is asymptomatic and self-limited. Symptomatic individuals can present with a mononucleosis-like illness similar to that caused by EBV. The illness lasts days to weeks and is characterized by fever, extreme fatigue, myalgia, mild pharyngitis, cough, nausea, diarrhea, and headache. Spiking fevers to 39° to 40°C are not uncommon and can last for more than 2 weeks. Fatigue and malaise may persist for 4 weeks or longer.12 Physical examination shows cervical or generalized lymphadenopathy. Rarely, hepatomegaly, splenomegaly, or rash may develop. Jaundice is uncommon. Hepatic enzymes are mildly elevated in 90% of patients with CMV mononucleosis and may remain abnormal for months. During pregnancy, laboratory abnormalities that suggest acute infection include lymphopenia or lymphocytosis associated with atypical lymphocytes, and thrombocytopenia.26
The differential diagnosis of CMV mononucleosis includes EBV mononucleosis, primary human immunodeficiency virus infection, and toxoplasmosis infection. CMV hepatitis must be differentiated from that caused by hepatitis virus A, B, or C. Mononucleosis caused by CMV is less likely to cause severe pharyngitis, tonsillitis, or significant splenomegaly, and it does not produce heterophil antibodies compared with EBV-induced mononucleosis.48
Laboratory Diagnosis
The advantages and limitations of laboratory tests used to diagnose CMV are provided in Table 1. Laboratory studies are helpful adjuncts to clinical findings but, unfortunately, may not conclusively discriminate between primary and recurrent infection. Laboratory diagnosis of CMV infection relies on serologic evidence of infection and direct identification of CMV. Discrimination of primary from recurrent CMV is based on maternal CMV-specific serologies because isolation of the virus from urine, throat, or genital secretions can occur during both primary and recurrent disease. Determining the role of CMV in the clinical setting is complicated because many adults without a history of symptomatic disease are seropositive for CMV-immunoglobulin G (IgG), and intermittent shedding of CMV can occur for years after the primary infection and bear no relation to illness. Neither the presence of IgG antibody nor excretion of the virus defines recent infection. Thus, a combination of laboratory tests, interpreted within the clinical context, is needed for an accurate diagnosis.
TABLE 1. Tests Used to Detect CMV Infection*
| |||
Test | Specimens | Use | Limitations |
Antigen detection |
|
|
|
Viral culture | Urine | Virus detection | 3—4 wk for results |
| Blood | Virus for further study |
|
| Sterile tissue |
|
|
| Amniotic fluid |
|
|
Direct immunofluorescence | Urine | Rapid detection of virus (48 hr) | Limited culture still necessary |
| Blood |
|
|
| Amniotic fluid |
|
|
Nucleic acid amplification | Urine | Rapid detection of virus | Expensive |
| Blood | Quantification of viral load | False-positives due to contamination |
| CSF | Virus strain typing |
|
| Amniotic fluid |
|
|
Antibody detection |
|
|
|
IgG | Serum | Demonstration of previous infection | Seroconversion takes 2—3 wk, requires 2 specimens |
|
| Demonstration of recent infection by seroconversion |
|
IgM | Serum | Demonstration of acute infection | Seropositive for up to 2 yr after acute infection |
|
|
| Rare cross-reactivity with EBV |
Complement fixation (CF) | Serum | Demonstration of rising titer | As above for serology |
CMV, cytomegalovirus; CSF, cerebrospinal fluid; IgG, immunoglobulin G; IgM, immunoglobulin M; EBV, Epstein-Barr virus
(Adapted from Rawlinson WD: Diagnosis of human cytomegalovirus infection and disease. Pathology 31:110, 1999.)
Previously, a rise in anti-CMV antibody titer by complement fixation was used to diagnosis primary CMV infection. However, the more efficient and accurate enzyme-linked immunosorbent assay (ELISA) has replaced the complement fixation test. Serologic diagnosis relies on the identification of patterns of immunoglobulin M (IgM) and IgG consistent with primary infection. The presence of IgM, the initial antibody response to infection, provides evidence of primary infection but must be interpreted with caution because anti-CMV IgM can be detected for up to 2 years in 5% of individuals after primary infection. Reinfection and reactivation of virus is associated with a positive IgM and/or a rise in complement fixing antibody titer in approximately 30% of cases. Occasionally, biologic false-positives can occur from cross-reactivity with antibodies related to EBV or autoimmune disorders.49 On average, detection of CMV-specific IgM antibody by ELISA has a sensitivity of only 75% for primary infection. This leaves 25% of women with primary CMV without detectable levels of IgM and a false-negative result.50
Demonstration of IgG antibody seroconversion or a fourfold increase in CMV-IgG antibody titer on samples collected 2 to 4 weeks apart and tested at the same time often is considered the gold standard to diagnose primary CMV infection but, like IgM determinations, is not always accurate. In some cases, rise in the antibody titer may be delayed up to 4 weeks from the time of acute illness.51
Avidity tests can be used to supplement ELISA. Low-avidity IgG antibodies are produced early in the infection and can be used as another marker of recent primary infection. The match between the host IgG antibodies and the target antigens improves as the host reacts to a pathogen. This characteristic has been used to date the acute infection using an avidity index (AI).52 An AI of less than 30% is highly suggestive of a recent primary infection (less than 3 months). An AI between 30% and 50% is possibly related to a recent infection, and an AI of greater than 50% is inconsistent with a recent primary infection.53 Avidity tests generally are used as an adjunct to ELISA because they are technically more difficult to perform and labor intensive.54
Diagnosis of recurrent CMV infection depends on viral isolation from urine, saliva, or cervix rather than serologic techniques because antibody levels do not usually change in an immunocompentent host. Direct viral detection is accomplished by viral culture, nucleic acid amplification, direct fluorescent antibody test, and, rarely, by detection of pp65 antigen in the blood.49
Viral culture can be performed on urine, buffy coat of peripheral cells, bronchoalveolar lavage specimens, nasopharyngeal aspirates, throat washings, semen, cervical and vaginal secretions, stool, tears, amniotic fluid, and breast milk. Specimens for culture should be inoculated onto cells as soon as possible after collection, preferably within 4 hours. Samples taken after-hours or samples shipped to other laboratories for culture can be stored at 4°C for up to 1 week but have reduced yields. At 10 to 14 days after inoculation, a cytopathic effect is observed in infected cells, although 21 to 28 days of incubation is needed to ensure a negative result. Unfortunately, viral culture is difficult, expensive, and time-consuming, taking up to 4 weeks for definitive results. The length of time necessary for diagnosis by culture can interfere with clinical decision making, particularly in the case of pregnancy.
Direct fluorescent antibody testing provides more rapid detection of CMV. This technique relies on limited culture of virus, but viral antigens can be detected on the surface of cells within 48 hours.55 The virus is centrifuged onto cell culture monolayers maintained in small shell-vials under coverslips and incubated for 24 to 48 hours. A pool of monoclonal antibodies to immediate early and early antigens and a fluorescein conjugated secondary antibody are added and the foci of infection identified with a fluorescence microscope.56 Compared with viral culture, this method has a sensitivity of 80% to 90% and a specificity of 95% to 100%.
Nucleic acid amplification by polymerase chain reaction (PCR) is the newest and most promising rapid method to detect virus. This method requires specific primers complementary to DNA sequences on either side of a target CMV DNA segment. A single gene copy may be amplified up to 1 million fold.57 PCR has a sensitivity of 80% to 100% when compared with that of culture; however, this high sensitivity can lead to false-positive results based on the amplification of extraneous viral contaminants or defective, noninfectious viral particles. PCR also can be used to quantify levels of virus.49
In summary, primary and recurrent CMV infection only can be distinguished by serologic studies. Seroconversion of CMV-specific IgG antibodies from negative to positive is the hallmark of primary infection but, unfortunately, preinfection specimens are rarely available in routine clinical practice. CMV IgG antibody can indicate either a past or current infection, and a single high titer is not helpful to determine the difference. A rise from a low titer to a very high titer on two serum samples obtained 2 to 3 weeks apart suggests a recent primary infection but is not diagnostic because recurrent infection also can boost the antibody titer. The presence of CMV-IgM antibody suggests a recent primary CMV infection,58 but, again, results must be interpreted with caution because IgM antibody may persist 10 to 12 weeks or longer after primary infection.59
CONGENITAL INFECTION
Congenital CMV infection occurs by hematogenous spread to the placenta followed by placental infection and subsequent fetal infection. Transmission can occur anytime during pregnancy. The influence of gestational age at the time of maternal acquisition of CMV infection is not as clear as with other congenital infections. However, the worst fetal sequelae have been associated with infection in the first trimester,26,48 and clinically significant handicaps are associated more often with primary infection before 27 weeks' gestation.35 Infants born to mothers infected early in pregnancy also are more likely to be small for gestational age and to have microcephaly and intracranial calcifications, whereas infants born to mothers infected later in pregnancy are more likely to have hepatitis, pneumonia, purpura, and severe thrombocytopenia.60 As noted previously, primary maternal infection is more likely to result in intrauterine transmission than recurrent infection.
Prenatal Diagnosis
Despite the common occurrence of primary maternal CMV infection and the significant risk of sequelae for offspring of infected mothers, routine maternal prenatal screening has not been advocated because a reliable and easy method to diagnose fetal infection is not available. Unfortunately, patients often must make decisions regarding pregnancy management based on empiric risk figures only.
Because the majority of primary and recurrent infections are asymptomatic, the diagnosis of CMV infection in pregnancy often is missed. Suspicion of maternal infection should be confirmed by appropriate serologic tests because ascertainment of primary versus recurrent infection is critical to predict neonatal prognosis. Seroconversion or rising titers of IgG or IgM often are difficult to show, as most women do not know their prepregnancy CMV serostatus. Paired samples run 1 to 4 weeks apart may show rising titers, but the time necessary for this diagnostic test prevents a rapid clinical decision. IgG avidity testing may assist in the diagnosis of primary CMV, but these tests generally are only available at research laboratories.
Direct diagnosis of fetal infection is difficult and no consensus exists regarding a gold standard for diagnosis. Clinical judgment is at the core of accurate diagnosis. Integration of maternal symptoms, physical findings and serology together with amniotic fluid culture and/or PCR results, fetal IgM determination, and ultrasound findings form the foundation of prenatal diagnosis. Unfortunately, definitive fetal diagnosis often can be made only at autopsy or delivery.
ISOLATION OF CYTOMEGALOVIRUS FROM AMNIOTIC FLUID.
The first prenatal diagnosis of fetal CMV infection occurred in 1971 by isolation of the virus from amniotic fluid.61 Because the renal tubular epithelium is a major site of viral replication, the infected fetus presumably excretes CMV into amniotic fluid via fetal urine.62 Studies have documented the utility of amniotic fluid cultures to detect fetal infection, and many consider amniotic fluid culture to be the optimal site to establish a prenatal diagnosis of congenital infection.63,64 Testing the amniotic fluid after 20 weeks' gestation is recommended to reduce the number of false-negative culture results. Sensitivity of amniotic fluid CMV culture ranges from 80% to 100%,64,65,66 and specificity is close to 100%.65 Recently, PCR of amniotic fluid has been added as a rapid means of detection, but results must be interpreted with caution because of possible false-positives.
CHORIONIC VILLUS SAMPLING.
Chorionic villus sampling (CVS) has been proposed as an option to diagnose congenital CMV in the first trimester. Diagnosis of fetal CMV infection in the first trimester would allow earlier, safer termination of pregnancy if desired by the patient. Because the virus seems to infect the placenta initially with subsequent amplification and passage to the fetus,67 placental tissue theoretically offers a potential source to detect fetal infection. However, limited data exist on the sensitivity and specificity of culture or PCR of placental tissue or both. Hogge and colleagues68 performed CVS on four of six pregnancies complicated by primary maternal CMV infection. All four had negative PCR results that were confirmed at birth with negative neonatal urine cultures. More research is needed before CVS is routinely used for first-trimester diagnosis of CMV infection.
CORDOCENTESIS FOR FETAL SEROLOGY.
The fetal immune system is capable of a consistent response to certain antigenic stimuli by 22 weeks' gestation, and evaluation of fetal sera for organism-specific IgM has been used to diagnose certain congenital infections. However, production of specific IgM appears to be related to the infecting organism. For example, although the majority of fetuses with congenital rubella infection produce specific IgM after 22 weeks,69 only 15% of fetuses with congenital toxoplasmosis produce specific IgM between 24 and 29 weeks.70 Lynch and coworkers71 found that although total IgM levels were elevated in infected fetuses sampled at 21 and 23 weeks' gestation, CMV-specific IgM was present in only one of five fetuses tested. This is in contrast to a prevalence of 50% to 95% among infected neonates72 and likely represents immaturity of the fetal immune system at this gestational age.
Despite the variability of fetal production of IgM, CMV-specific IgM determinations by ELISA can be attempted on fetal blood obtained by cordocentesis. To increase the sensitivity of this assay and to eliminate false-positives due to rheumatoid factor, sera should be preabsorbed with a modified staphylococcal protein A preparation.50 The few studies performed on the diagnostic utility of fetal CMVspecific IgM have had variable results. Hogge and coworkers68 performed cordocentesis along with amniocentesis on three pregnant women with primary maternal CMV infection. Compared with neonatal culture, all three cordocentesis results were incorrect with two false-negatives and one false-positive at 22.5 to 25 weeks. In contrast, Hohlfeld and coworkers64 reported only two false-negatives and no false-positives in 16 patients evaluated. Notably, in both series, the diagnosis was accurately made by amniocentesis, and fetal IgM determinations added little further information. The true sensitivity and specificity of fetal IgM determination are not known, but sensitivity has been between 69% and 75% in small series.65,73 Based on these findings, it is evident that fetal IgM alone cannot be used to exclude or confirm congenital CMV infection.
Although fetal IgM determination may not add useful diagnostic information, fetal blood obtained at cordocentesis may yield prognostic information if sent to evaluate hematologic and hepatic parameters. Extreme thrombocytopenia, anemia, or elevated liver function test results documented in fetal blood suggest more severe disease; however, how these parameters relate to childhood handicaps remains unknown.64
ULTRASOUND.
Often, congenital CMV infection is suspected because of abnormal findings on routine obstetric ultrasound. Ventricular dilation, microcephaly, cerebral calcifications, and hydrops are considered suspicious for CMV infection. This information is derived from case series that evaluated fetuses of mothers with documented primary CMV or fetuses with abnormal findings on routine ultrasound that led to a workup for congenital infection. Ultrasound findings associated with CMV infection are diverse and can change over time in a given fetus. For example, ascites or pericardial or pleural effusions can resolve; however, this does not necessarily predict an improved prognosis.62,71 Any fetus with evidence of unexplained hydrops should be evaluated for CMV.71 Abnormalities such as ventricular dilation, oligohydramnios,71 pericardial effusions,62 hydrops, and ascites documented by ultrasound have been associated with more severe syndromes.64
LIMITATIONS OF PRENATAL DIAGNOSIS.
Although amniotic fluid culture is sensitive, its use as the sole mode of diagnosis is problematic. PCR applied to amniotic fluid samples further increases the sensitivity of amniotic fluid analysis and provides a more rapid diagnosis. However, because of reports of false-negative amniotic fluid culture and false-positive amniotic fluid PCR results, a combination of amniocentesis and fetal blood sampling may provide the optimal means to diagnose fetal CMV infection.
Cordocentesis also provides fetal blood for testing to determine fetal prognosis. Hematologic evidence of systemic infection appears associated with a worse prognosis.74 Similarly, abnormalities on ultrasound suggest a poor prognosis; however, several cases in the literature indicate that systemic fetal CMV does not universally result in a severely handicapped neonate.63,75,76 Thus, counseling the mother regarding a possible poor prognosis for her infant must be framed appropriately. Further study is necessary to define fetal indicators predictive of severe handicap.
Neonatal Diagnosis of Congenital Infection
At birth, the majority of CMV-infected infants are asymptomatic and only about 10% show signs of CID. Clinical features of CID most frequently include hepatosplenomegaly, microcephaly, jaundice, and petechiae. Hepatosplenomegaly is caused by extramedullary hematopoiesis and mild hepatitis, which usually is associated with elevated liver enzymes and direct and indirect hyperbilirubinemia. Petechiae are the result of thrombocytopenia and usually resolve within a few weeks to months. Other less-common findings include purpura, pneumonia, chorioretinitis, cerebral calcifications, hemolytic anemia, and inguinal hernias in males.13
Because the signs and symptoms of symptomatic CMV infection in newborns so often overlap with those of other neonatal diseases, a definitive diagnosis requires laboratory testing. Viral isolation from infants within the first 3 weeks of life is considered proof of congenital infection and the most sensitive means of diagnosis. The usual site for isolation is the urine, but the virus has also been recovered from cerebrospinal fluid, saliva, buffy coat, and biopsy specimens or autopsy tissue.77
Prognosis
The incidence and severity of sequelae in congenitally infected infants are related to the presence or absence of symptoms at birth and to the stage (primary or recurrent) of maternal CMV infection experienced during pregnancy.
SYMPTOMATIC VERSUS ASYMPTOMATIC INFECTION AT BIRTH.
Table 2 presents the incidence of sequelae in symptomatic infants compared with asymptomatic infants at birth. Prognosis is poor for infants with CID at birth. More than 90% of symptomatic infants experience serious sequelae such as death (5.8%), chorioretinitis (20.4%), sensorineural hearing loss (58%), and mental retardation (55%). Boppana and coworkers,78 based on data from 106 neonates with symptomatic congenital CMV infection, confirmed the multisystem nature of the disease and its associated morbidity and mortality. Clinical and laboratory abnormalities indicated involvement of the heptobiliary, hematopoietic, and central nervous systems in the majority of infants studied in the first month of life. Greater than two thirds of these infants had at least one neonatal sign of central nervous system disease (e.g., microcephaly, lethargy/hypotonia, poor suck, seizures). These signs have been found to correlate with mental retardation in school-age children.74
TABLE 2. Sequelae in Children After Congenital CMV Infection According to Symptoms at Birth* and Type of Maternal Infection†
Sequela | Symptomatic % (No.) | Asymptomtic % (No.) | Primary % (No.) | Recurrent % (No.) |
Sensoneural hearing loss | 58.0 (58/100) | 7.4 (22/299) | 15.0 (18/120) | 5.0 (3/56) |
Bilateral hearing loss | 370.0 (37/100) | 2.7 (8/299) | 8.0 (10/120) | 0 (0/56) |
Speech threshold | 27.0 (27/100) | 1.7 (5/299) | 8.0 (9/120) | 0 (0/56) |
Chorioretinitis | 20.4 (19/93) | 2.5 (7/281) | 13.0 (9/68) | 0 (0/32) |
IQ < 70 | 55.0 (33/60) | 3.7 (6/159) | 6.0 (7/112) | 2.0 (1.54) |
Microcephaly, seizures, or paralysis | 51.9 (54/104) | 2.7 (9/330) | 6.0 (8/125) | 2.0 (1/64) |
Microcephaly | 37.5 (39/104) | 1.8 (6/330) | 5.0 (6/125) | 2.0 (1/64) |
Seizures | 23.1 (24/104) | 0.9 (3/330) | 5.0 (6/125) | 0 (0/64) |
Paresis/Paralysis | 12.5 (13/104) | 0 (0/330) | 1.0 (1/125) | 0 (0/64) |
Death§ | 5.8 (6/104) | 0.3 (1/330) | 2.0 (3/125) | 0 (0/64) |
IQ, intelligence quotient.
*Data from Stagno S, Whitley RJ: Herpesvirus infections in neonates and children: Cytomegalovirus and herpes simplex virus. In Holmes KK (ed): Sexually Transmitted Diseases, p 1197, McGraw Hill, 1999.
†Data from Fowler KB, Stagno S, Pass RF et al: The outcome of congenital cytomegalovirus infection in relation to maternal antibody status. N Engl J Med 326:665, 1992.
‡For the ear with better hearing.
§After the newborn period.
Mortality in the most severely affected infants can be as high as 30%. Death within the first month of life usually is caused by multiorgan system disease that produces severe hepatic dysfunction, disseminated intravascular coagulation, and secondary infections. Among the infants studied by Boppana and coworkers,78 12% died during the first 6 weeks of life. Death occurring outside this period but within 1 year of life usually is caused by progressive liver failure and failure to thrive. Death after 1 year of life usually occurs from malnutrition and infection among those with central nervous system infection and neurologic disability.79
By contrast, prognosis is relatively good for congenitally infected infants who are asymptomatic at birth. Only 5% to 15% of asymptomatic congenitally infected infants have long-term sequelae develop that become apparent within the first 2 years of life. Sensorineural hearing loss is the most common sequelae in these infants, occurring at an incidence of approximately 7.5% of infected infants asymptomatic at birth.80,81,82,83,84 The impairment is bilateral in nearly half of the cases and is of sufficient severity to impair verbal communication and learning. Other neurologic complications such as mental retardation, microcephaly, seizures, and paralysis nevertheless occur in approximately 2% to 3% of congenitally infected infants who were asymptomatic at birth.85
In summary, the likelihood that infants who survive symptomatic congenital CMV infection will have normal intellectual development and hearing is small.1 In contrast, the prognosis for infected infants who are asymptomatic at birth is good.
PRIMARY VERSUS RECURRENT MATERNAL INFECTION.
Most infants with reported sequelae have been born to mothers with primary infection during pregnancy.51 Congenital CMV infections in children of women with immunity acquired before pregnancy are less likely to be symptomatic at birth than those resulting from a primary infection.33,35
These children also are less likely to have severe sequelae (see Fig. 1). Fowler and coworkers51 compared the outcomes of 125 CMV-infected infants born to mothers who acquired primary CMV infection during pregnancy with 64 CMV-infected infants born to mothers with recurrent disease. The findings, presented in Table 2, indicate that pre-existing maternal antibody to CMV protects the fetus and lessens the severity of the sequelae of congenital CMV infection. Children infected after primary maternal infection were more likely to have functionally important sensorineural hearing loss and more likely to have intelligence quotient scores less than 70 than children infected after recurrent maternal infection. The children in the primary group also were more likely to be severely damaged as indicated by the prevalence of multiple sequelae (6%) in this group compared with that in the recurrent group (0%).
MANAGEMENT OF MATERNAL/CONGENITAL INFECTION
Counseling
Counseling a woman with primary CMV infection in pregnancy is complex, and a rational prenatal protocol to evaluate the fetus is complicated by vagaries in diagnostic testing and the lack of a consistent fetal prognostic indicator. A risk-based approach is recommended. A priori, the risk of a severely affected infant after a maternal primary CMV infection is 3% to 6% based on a 40% chance of congenital infection, a 10% to 15% chance of having an infant symptomatic at birth, and a 90% chance of sequelae among symptomatic neonates as outlined in Figure 1.
If diagnostic testing confirms congenital infection, the risk of severe neurologic damage in the infant increases to approximately 10%. Conversely, the remaining 90% of infants have an excellent prognosis. Therefore, for 60% to 70% of women with primary CMV during pregnancy, prenatal fetal testing yields a reassuring negative result and prevents unnecessary pregnancy termination. The remaining 30% to 40% of women will have a positive result. In utero prediction of which 10% of these fetuses will be severely affected is problematic. Serial ultrasound assessment and possibly cordocentesis at 22 weeks to evaluate the fetus for hematologic or hepatic abnormalities as prognostic indicators should be discussed. Some of these women will elect for termination rather than wait for further information provided by ultrasound or cordocentesis.68 If cordocentesis shows severe thrombocytopenia, platelet transfusion should be considered along with serial fetal blood samplings to document resolution. If fetal thrombocytopenia persists, mode of delivery may be altered.75
The significance of fetal infection in the absence of ultrasonographic signs and abnormal laboratory values remains unclear, but given the high percentage of asymptomatic congenital CMV infections at birth, it is reasonable to allow these pregnancies to proceed with serial ultrasound monitoring. At birth, all infected infants should receive close follow-up, including serial assessment of hearing and psychomotor development for several years.64
Treatment
There is no known treatment for CMV infection in pregnancy. Two antiretroviral agents, ganciclovir and foscarnet, are available to treat severe life-threatening or sight-threatening CMV infections in immunocompromised patients. Systemic ganciclovir treatment has not yielded significant clinical benefit for congenitally infected infants and is associated with significant toxicity.86,87,88 The variable nature of congenital CMV and the irreversible damage that occurs prenatally make postnatal treatment trials difficult. A controlled clinical trial of ganciclovir for treatment of congenital CMV is underway.79
Vaccine
Infants born to women with primary CMV infection during pregnancy are more likely to be seriously ill at birth, to have bilateral hearing loss, and to be mentally retarded than infants born to women with recurrent CMV infection during pregnancy.51 Unlike primary CMV infection, only a small percentage of women with recurrent infection transmit CMV to their infants, and these infants rarely experience severe sequelae.33,51 Therefore, vaccination of susceptible women to prevent primary CMV infection in pregnancy may provide protection against the damage from congenital CMV infection.89
Two live attenuated vaccines (AD-169 and Towne strains) have undergone efficacy trials in immune and nonimmune healthy hosts and renal transplant recipients.90,91,92 Among kidney transplant patients immunized with the Towne strain vaccine, morbidity associated with primary CMV infection was curtailed, but infection was not prevented.93 More important, however, the vaccine virus did not reactivate in immunocompromised hosts. A vaccine to prevent primary infection among pregnant women ideally should not induce latency or reactivate during pregnancy. Because of this theoretical potential, live attenuated vaccines generally are avoided in pregnancy.
Although research should continue on a live attenuated virus, a safer approach involves a subunit vaccine that provokes antibody formation to a viral envelope protein. Gylcoprotein gB is the most abundant protein complex of the virus envelope expressed on the surface of infected cells. Neutralizing antibody is present soon after infection and persists during the convalescent phase.94 Early trials in humans currently are underway.79
Prevention
In the absence of a suitable vaccine, the only measure available at this time to prevent CMV infection is to avoid exposure. CMV is not very contagious, and horizontal transmission requires close contact with infected secretions or occasionally fomites. The principal sources of exposure for women of childbearing age are sexual contacts and children secreting the virus. Behavioral intervention trials focused on improved hygiene among women with young children failed to provide a statistically significant difference in seroconversion rates among nonpregnant women. However, of the 14 pregnant women given the education intervention, none seroconverted over the 9-month follow-up period. This suggests that education regarding basic hygiene measures may decrease primary infection acquisition among pregnant women with young children at home.95 Pregnant women also should be advised to avoid close contact with individuals likely to shed CMV such as adults with symptoms consistent with mononucleosis and toddlers attending group day care.
Prenatal screening for CMV antibody remains controversial. Some investigators recommend screening women of childbearing age to determine antibody status. Seronegative women could then take precautions to avoid CMV infection such as avoidance of sexual contact with persons likely to shed CMV, careful hand washing after contact with children's secretions, and avoidance of shared eating utensils.95,96,97 This recommendation may not be cost-beneficial, however, because the efficacy of prevention counseling has not been definitively proved.98 Alternatively, basic hygiene practices could be recommended to all pregnant women without testing for CMV antibody status.
Currently, routine CMV screening during pregnancy to identify those who have acquired primary infection during pregnancy is not recommended because of the low (1% to 2%) incidence of primary CMV infection in pregnancy and the relatively low (40%) likelihood that congenital infection occurs. Further, the invasive and uncertain nature of fetal testing, the lack of definitive prognostic indicators, and the lack of effective antiviral therapy make routine serologic testing in pregnancy of limited value.
ACKNOWLEDGMENT
This work was supported under a cooperative agreement from the Centers for Disease Control through the Association of Teachers of Preventive Medicine.
REFERENCES
Alford CA, Stagno S, Pass RF et al: Congenital and perinatal cytomegalovirus infections. Rev Infect Dis 12: S745, 1990 |
|
Weller TH: The cytomegaloviruses: Ubiquitous agents with protean clinical manifestations. N Engl J Med 285: 267, 1971 |
|
Daniel Y, Gull I, Peyser R et al: Congenital cytomegalovirus infection. Eur J Obstet Gynecol Reprod Biol 63: 7, 1995 |
|
Gold E, Nankervis GA: Cytomegalovirus. In Evans AS (ed): Viral Infections in Humans: Epidemiology and Control, pp 143–161. New York, Elsevier, 1976 |
|
Chee MS, Bankier AT, Beck S et al: Analysis of the protein-coding content of the sequence of human cytomegalovirus strain AD 169. Curr Top Microbiol Immunol 154: 125, 1990 |
|
Smith JD, DeHarven E: Herpes simplex virus and human cytomegalovirus replication in WI-38 cells. I. Sequence of viral replication. J Virol 12: 919, 1978 |
|
Griffiths PD, Grundy JE: Molecular biology and immunology of cytomegalovirus. Biochem J 241: 313, 1987 |
|
Sinclair J, Sissons JGP: Human cytomegalovirus: Pathogenesis and models of latency. Semin Virol 5: 249, 1994 |
|
Mathijs JM, Rawlinson WD, Bilous AM et al: Cellular localization of human cytomegalovirus reactivation in the cervix. J Infect Dis 163: 921, 1991 |
|
Mocarski ES: Cytomegalovirus biology and replication. In Roizman B, Whitley C, Lopez C (eds): The Human Herpesviruses. New York, Raven Press, 1993 |
|
Honess RW, Roizman B: Regulation of herpesvirus macromolecular synthesis. I. Cascade regulation of the synthesis of three groups of viral proteins. J Virol 14: 8, 1974 |
|
Nesmith DJ, Pass RF: Cytomegalovirus infection in adolescents. Adolesc Med 6: 79, 1995 |
|
Stagno S: Cytomegalovirus. In Remington JS, Klein JD (eds): Infectious Diseases of the Fetus and Newborn Infant, pp 312–353. 4th ed. Philadelphia, WB Saunders, 1995 |
|
Van der Meer JT, Drew WL, Bowden RA et al: Summary of the International Consensus Symposium on Advances in the diagnosis, treatment and prophylaxis of cytomegalovirus infection. Antiviral Res 32: 119, 1996 |
|
Stagno S, Reynolds D, Tsiantos A et al: Cervical cytomegalovirus excretion in pregnant and non-pregnant women: Suppression in early gestation. J Infect Dis 131: 522, 1975 |
|
Lang DJ, Kummer JF: Cytomegalovirus in semen: Observations in selected populations. J Infect Dis 132: 472, 1975 |
|
Weller TH, Hanshaw JB: Virologic and clinical observations on cytomegalic inclusion disease. N Engl J Med 266: 1233, 1962 |
|
Chandler SH, Alexander ER, Holmes KK: Epidemiology of cytomegalovirus infection in a heterogeneous population of pregnant women. J Infect Dis 152: 249, 1985 |
|
Stagno S, Cloud GA: Working parents: The impact of day care and breast-feeding on cytomegalovirus infections in offspring. Proc Natl Acad Sci USA Mar 29;91:2384, 1994 |
|
Pass RF, August AM, Dworsky M et al: Cytomegalovirus infection in day care centers. N Engl J Med 307: 477, 1982 |
|
Adler SP: The molecular epidemiology of cytomegalovirus: Evidence for viral transmission among children attending a day care center. J Infect Dis 152: 760, 1985 |
|
Murph JR, Bale JF Jr, Murray JC et al: Cytomegalovirus transmission in a Midwest day care center: Possible relationship to child care practices. J Pediatr 109: 35, 1986 |
|
Adler SP: Molecular Epidemiology of cytomegalovirus: Evidence for viral transmission to parents from children infected at a day care center. Pediatr Infect Dis J 5: 315, 1986 |
|
Hutto C, Little EA, Ricks R et al: Isolation of cytomegalovirus from toys and hands in a daycare center. J Infect Dis 154: 527, 1986 |
|
Faix RG: Survival of cytomegalovirus on environmental surfaces. J Pediatr 106: 649, 1985 |
|
Brown HL, Abernathy MP: Cytomegalovirus infection. Semin Perinatol 22: 260, 1998 |
|
Yeager AS, Palumbo PE, Malachowski N et al: Sequelae of maternally derived cytomegalovirus infections in premature infants. J Pediatr 102: 918, 1983 |
|
Betts RR: Cytomegalovirus infection epidemiology and biology in adults. Semin Perinatol 7: 22, 1983 |
|
Yow MD, Williamson DW, Leeds LF et al: Epidemiologic characteristics of cytomegalovirus infection in mothers and their infants. Am J Obstet Gynecol 158: 1189, 1988 |
|
Peckham CS, Chin KS, Coleman JC et al: Cytomegalovirus infection in pregnancy: Preliminary findings from a prospective study. Lancet 1: 1352, 1983 |
|
Grant S, Edmond E, Syme J: A prospective study of cytomegalovirus infection in pregnancy: Laboratory evidence of congenital infection following maternal primary and reactivated infection. J Infect 3: 24, 1981 |
|
Griffiths P, Baboonian C, Ashby D: The demographic characteristics of pregnant women infected with cytomegalovirus. Int J Epidemiol 14: 447, 1985 |
|
Stagno S, Pass RF, Dworsky ME et al: Congenital cytomegalovirus infection: The relative importance of primary and recurrent maternal infection. N Engl J Med 306: 945, 1982 |
|
Ahlfors K, Ivarsson SA, Hohnsson T et al: Primary and secondary maternal cytomegalovirus infections and their relation to congenital infection: Analysis of maternal sera. Acta Paediatr Scand 71: 109, 1982 |
|
Stagno S, Pass RF, Cloud G et al: Primary cytomegalovirus infection during pregnancy: Incidence, transmission to the fetus and clinical outcome in two populations of different socioeconomic backgrounds. JAMA 256: 904, 1986 |
|
Gibbs RS, Sweet RL: Maternal and fetal infectious disorders. In Creasy RK, Resnik R (eds): Maternal-Fetal Medicine, pp 682–688. 4th ed. Philadelphia, WB Saunders, 1999 |
|
Stagno S, Cloud G, Pass RF et al: Factors associated with primary cytomegalovirus infection during pregnancy. J Med Virol 13: 347, 1984 |
|
Griffiths PD, Campbell-Benzie A, Heath RB: A prospective study of primary cytomegalovirus infection in pregnant women. Br J Obstet Gynaecol 87: 308, 1980 |
|
Dobbins JG, Adler SP, Pass RF et al: The risks and benefits of cytomegalovirus transmission in child day care. Pediatrics 94: 1016, 1994 |
|
Pass RF, Hutto C, Ricks R et al: Increased rate of cytomegalovirus infection among parents of children attending day-care centers. N Engl J Med 34: 1414, 1986 |
|
Adler SP: Cytomegalovirus and child day-care: Risk factors for maternal infection. Pediatr Infect Dis J 10: 590, 1991 |
|
Adler SP: Cytomegalovirus and child day care: Evidence for an increase infection rate among day-care workers. N Engl J Med 321: 1290, 1989 |
|
Yeager AS: Longitudinal, serological study of cytomegalovirus infections in nurses and in personnel without patient contact. J Clin Microbiol 2: 448, 1975 |
|
Friedman HM, Lewis MR, Nemerofsky DM et al: Acquisition of cytomegalovirus infection among female employees at a pediatric hospital. Pediatr Infect Dis 3: 233, 1984 |
|
Dworsky ME, Welch K, Cassady G et al: Occupational risk for primary cytomegalovirus infection among pediatric health-care workers. N Engl J Med 309: 950, 1983 |
|
Balfour CL, Balfour HH Jr: Cytomegalovirus is not an occupational risk for nurses in renal transplant and neonatal units. JAMA 256: 1909, 1986 |
|
Raynor BD: Cytomegalovirus infection in pregnancy. Semin Perinatol 17: 394, 1993 |
|
Demmler GJ: Cytomegalovirus. In Gonik B (ed): Viral Diseases in Pregnancy, pp 69–91. New York, Springer-Verlag, 1994 |
|
Rawlinson WD: Diagnosis of human cytomegalovirus infection and disease. Pathology 31: 109, 1999 |
|
Stagno S, Tinker MK, Elrod C et al: Immunoglobulin M antibodies detected by enzyme-linked immunosorbent assay and radioimmunoassay in the diagnosis of cytomegalovirus infections in pregnant women and newborn infants. J Clin Microbiol 21: 930, 1985 |
|
Fowler KB, Stagno S, Pass RF et al: The outcome of congenital cytomegalovirus infection in relation to maternal antibody status. N Engl J Med 326: 663, 1992 |
|
Grangeot-Keros L, Mayaux M, Lebon P et al: Value of cytomegalovirus (CMV) IgG avidity index for the diagnosis of primary CMV infection in pregnant women. J Infect Dis 175: 944, 1997 |
|
Grangeot-Keros L, Simon B, Audibert F et al: Should we routinely screen for cytomegalovirus antibody during pregnancy? Intervirology 41: 158, 1998 |
|
Blackburn NK, Besselaar TG, Schoub BD et al: Differentiation of primary cytomegalovirus infection from reactivation using the urea denaturation test for measuring antibody avidity. J Med Virol 33: 6, 1991 |
|
Griffiths PD, Panjwani DD, Stirk PR et al: Rapid diagnosis of cytomegalovirus infection in immunocompromised patients by detection of early antigenic fluorescent foci. Lancet 2: 1242, 1984 |
|
Paya CV, Smith TF, Ludwig J et al: Rapid shell vial culture and tissue histology compared with serology for the rapid diagnosis of cytomegalovirus infection in liver transplantation. Mayo Clin Proc 64: 670, 1989 |
|
Pillay D, Griffiths P: Diagnosis of cytomegalovirus infection: A review. Genitourin Med 68: 183, 1992 |
|
Griffiths PD, Stagno S, Pass RF et al: Infection with cytomegalovirus during pregnancy: Specific IgM antibodies as a marker of recent primary infection. J Infect Dis 145: 647, 1982 |
|
Demmler GJ, Six HR, Hurst SM et al: Enzyme-linked immunosorbent assay for the detection of IgM-class antibodies to cytomegalovirus. J Infect Dis 153: 1152, 1986 |
|
Yow MD: CMV infection in young women. Hosp Pract 30: 61, 1990 |
|
Davis LE, Tweed GV, Chin TDY et al: Intrauterine diagnosis of cytomegalovirus infection: Viral recovery from amniocentesis fluid. Am J Obstet Gynecol 109: 1217, 1971 |
|
Weiner CP, Grose C: Prenatal diagnosis of congenital cytomegalovirus infection by virus isolation from amniotic fluid. Am J Obstet Gynecol 163: 1253, 1990 |
|
Grose C, Weiner CP: Prenatal diagnosis of congenital cytomegalovirus infection: Two decades later. Am J Obstet Gynecol 163: 447, 1990 |
|
Hohlfeld P, Vial Y, Maillard-Brignon C et al: Cytomegalovirus fetal infection: Prenatal diagnosis. Obstet Gynecol 78: 615, 1991 |
|
Donner C, Liesnard C, Content J et al: Prenatal diagnosis of 52 pregnancies at risk for congenital cytomegalovirus infection. Obstet Gynecol 82: 481, 1993 |
|
Lamy H, Mulongo K, Gadisseux JF et al: Prenatal diagnosis of fetal cytomegalovirus infection. Am J Obstet Gynecol 166: 91, 1992 |
|
Goff E, Griffith BP, Booss J: Delayed amplification of cytomegalovirus infection in the placenta and maternal tissues during late gestation. Am J Obstet Gynecol 156: 1265, 1987 |
|
Hogge WA, Buffone GJ, Hogge JS: Prenatal diagnosis of cytomegalovirus (CMV) infection: A preliminary report. Prenatal Diagn 13: 131, 1993 |
|
Daffos F, Forestier F, Grangeot-Keros L et al: Prenatal diagnosis of congenital rubella. Lancet 2: 1, 1984 |
|
Daffos F, Forestier F, Capella-Pavlovsky M et al: Prenatal management of 746 pregnancies at risk for congenital toxoplasmosis. N Engl J Med 318: 271, 1988 |
|
Lynch L, Daffos F, Emanuel D et al: Prenatal diagnosis of fetal cytomegalovirus infection. Am J Obstet Gynecol 165: 714, 1991 |
|
Ahlfors K, Forsgren M Griffiths P et al: Comparison of four serological tests for the detection of specific immunoglobulin M in cord sera of infants congenitally infected with cytomegalovirus. Scand J Infect Dis 19: 303, 1987 |
|
Nicolini U, Kusterman A, Tassis B et al: Prenatal diagnosis of congenital human cytomegalovirus infection. Prenatal Diagn 14: 903, 1994 |
|
Conboy TJ, Pass RF, Stagno S et al: Early clinical manifestations and intellectual outcome in children with symptomatic congenital cytomegalovirus infection. J Pediatr 111: 343, 1987 |
|
Watt-Morse ML, Laifer SA, Hill LM: The natural history of fetal cytomegalovirus infection as assessed by serial ultrasound and fetal blood sampling: A case report. Prenatal Diagn 15: 567, 1995 |
|
Binder ND, Buckmaster JW, Benda GL: Outcome for fetus with ascites and cytomegalovirus infection. Pediatrics 82: 100, 1988 |
|
Feigin RD, McCracken GH Jr, Klein JO: Diagnosis and management of meningitis. Pediatr Infect Dis J 11: 785, 1992 |
|
Boppana SB, Pass RF, Britt WJ et al: Symptomatic congenital cytomegalovirus infection: Neonatal morbidity and mortality. Pediatr Infect Dis J 11: 93, 1992 |
|
Stagno S, Whitley RJ: Herpesvirus infections in neonates and children: Cytomegalovirus and herpes simplex virus. In Holmes KK, Sparling PF, Mardh P et al (eds): Sexually Transmitted Diseases, pp 1191–1212. 3rd ed. New York, McGraw Hill, 1999 |
|
Pass RR, Fowler KB, Boppana S: Progress in cytomegalovirus research. In Landini MP (ed): Proceedings of the Third International Cytomegalovirus Workshop, pp 3–10. Bologna, Italy, June 1991, London, Excerpta Medica, 1991 |
|
Kumar ML, Nankervis GA, Gold E: Inapparent congenital cytomegalovirus infection: A follow-up study. N Engl J Med 288: 1370, 1973 |
|
Saigal S, Lunyk O, Lance RP et al: The outcome in children with congenital cytomegalovirus infection: A longitudinal follow-up study. Am J Dis Child 136: 896, 1982 |
|
Melish ME, Hanshaw JB: Congenital cytomegalovirus infection: Developmental progress of infants detected by routine screening. Am J Dis Child 126: 190, 1973 |
|
Pearl KN, Preece PM, Ades A et al: Neurodevelopmental assessment after congenital cytomegalovirus infection. Arch Dis Child 61: 323, 1986 |
|
Williamson WD, Percy AK, Yow MD et al: Asymptomatic congenital cytomegalovirus infection: Audiologic, neuroradiologic, and neurodevelopmental abnormalities during the first year. Am J Dis Child 144: 1365, 1990 |
|
Nigro G, Scholz H, Bartman U: Ganciclovir therapy for symptomatic congenital cytomegalovirus infection in infants: A two-regimen experience. J Pediatr 124: 318, 1994 |
|
Pehlivanoglu E: Ganciclovir therapy for cytomegalovirus infection in infants. J Pediatr 125: 670, 1994 |
|
Whitley RJ, Cloud G, Gruber W et al: Ganciclovir treatment of symptomatic congenital cytomegalovirus infection: Results of a phase II study. National Institute of Allergy and Infectious Diseases Collaborative Antiviral Study Group. J Infect Dis 175: 1080, 1997 |
|
Yow MD, Demmler G: Congenital cytomegalovirus disease: 20 years is long enough. N Engl J Med 326: 702, 1992 |
|
Elk SD, Stern H: Development of a vaccine against mental retardation caused by cytomegalovirus infection in utero. Lancet 1: 1, 1974 |
|
Plotkin SA, Furnkawa T, Zygraich N et al: Candidate cytomegalovirus strain for human vaccination. Infect Immun 12: 521, 1975 |
|
Adler SP, Hempfling SH, Starr SE et al: Safety and immunogenicity of the Town strain cytomegalovirus vaccine. Pediatr Infect Dis J 17: 200, 1998 |
|
Adler SP: Current prospects for immunization against automegaloviral disease. Infect Agents Dis 5: 39, 1996 |
|
Britt WJ, Vugler L, Butfiloski EJ et al: Cell surface expression of human cytomegalovirus (HCMV) gp55–116(gB): Use of HCMV-recombinant vaccinia virus-infected cells in analysis of the human neutralizing antibody response. J Virol 64: 1079, 1990 |
|
Adler SP, Finney JW, Manganello AM et al: Prevention of child-to-mother transmission of cytomegalovirus by changing behaviors: A randomized controlled trial. Pediatr Infect Dis J 15: 240, 1996 |
|
Demmler GJ: Summary of a workshop on surveillance for congenital cytomegalovirus disease. Rev Infect Dis 13: 315, 1991 |
|
Yow MD: Congenital cytomegalovirus disease: A NOW problem. J Infect Dis 159: 163, 1989 |
|
Hagay ZJ, Biran G, Ornoy A et al: Congenital cytomegalovirus infection: A long-standing problem still seeking a solution. Am J Obstet Gynecol 174: 241, 1996 |



 60dB†
60dB†